This document serves as a guide to Speech and Language Therapists working with people with multiple system atrophy (MSA). It draws on available literature in MSA, Parkinson’s disease and other atypical Parkinsonism disorders. It does not cover aetiology, epidemiology, neuropathology and medical management in any depth. Further reading on these topics and others can be accessed via the list of resources (including the MSA website), given at the end of the document.
The Multiple System Atrophy Trust (MSA Trust) produces a series of specialist MSA factsheets for health professionals to enable them to improve the treatment people with MSA receive. Other factsheets can be found on our website: www.msatrust.org.uk
The Multiple System Atrophy Trust (MSA Trust) is the only charity working in the UK and Ireland specifically to support people with MSA. As well as helping people who have MSA, the Trust supports anyone affected by the disease, including carers, families, friends and health professionals.
The Trust employs MSA Health Care Specialists, manages a telephone and email advice service and runs a network of support groups. We provide up-to-date literature for people affected by MSA and for health professionals. We also fund vital research to find the cause, and one day, cure for MSA.
To ensure services are accessible to everyone, the Trust is committed to providing services for people affected by MSA free of charge. The MSA Trust is a charity funded entirely on voluntary donations.
The MSA Trust is always keen to receive feedback about the information we produce, please email support@msatrust.org.uk with any comments.
Introduction
MSA is a rare progressive neurological disorder that affects adult men and women and leads to premature death. Currently, there is no known cause or cure. MSA causes degeneration or atrophy of nerve cells in several (or multiple) areas of the brain which results in problems with movement, balance and autonomic functions of the body such as swallowing, bowel, bladder and blood pressure control.
Globally, around five people per 100,000 have MSA which equates to almost 3,000 people living with MSA in the UK [1]. Parkinson’s disease is about 45 times more common, affecting about 200 per 100,000 in the UK [2].
MSA usually starts between the ages of 50-60 years, but it can affect people younger and older. MSA does not appear to be hereditary although current research is examining whether there may be a genetic predisposition to develop the disease. It affects both sexes equally [7].
Neuropathology
MSA falls within the entity of the spectrum of alphasynucleinopathies and oligodendrogliopathies. The mechanisms underlying the condition and the factors that trigger MSA onset are yet to be established. Environmental and genetic influences have been cited [4] however definitive cause and risk factors are yet to be established. Symptoms of MSA are a manifestation of the accumulation of alpha-synuclein in glial inclusion bodies, originating in the striatonigral, olivopontocerebellar and central autonomic (brain stem) regions. People diagnosed with MSA will eventually present with an overlap of symptoms, as illustrated in Figure 1.
![Figure 1 - MSA signs and symptoms (Adapted from Swan [5])](https://www.msatrust.org.uk/wp-content/smush-webp/2026/03/msa-signs-and-symptoms.png.webp)
Clinical Diagnosis of MSA
The term MSA was first used in 1969 – prior to this it had previously been known as Shy -Drager Syndrome. The first diagnostic criteria for MSA were proposed in 1989 [7] and Second Consensus Criteria were defined in 2008 [8] which define three levels of certainty of the diagnosis—possible, probable and definite MSA. New Movement Disorder Society (MDS) criteria for MSA diagnosis were published in 2022 using an evidence-based and consensus-based methodology [54] . Neuropathologically established MSA criteria remain the same, but new categories of clinically established MSA, clinically probable MSA and possible prodromal MSA are proposed.
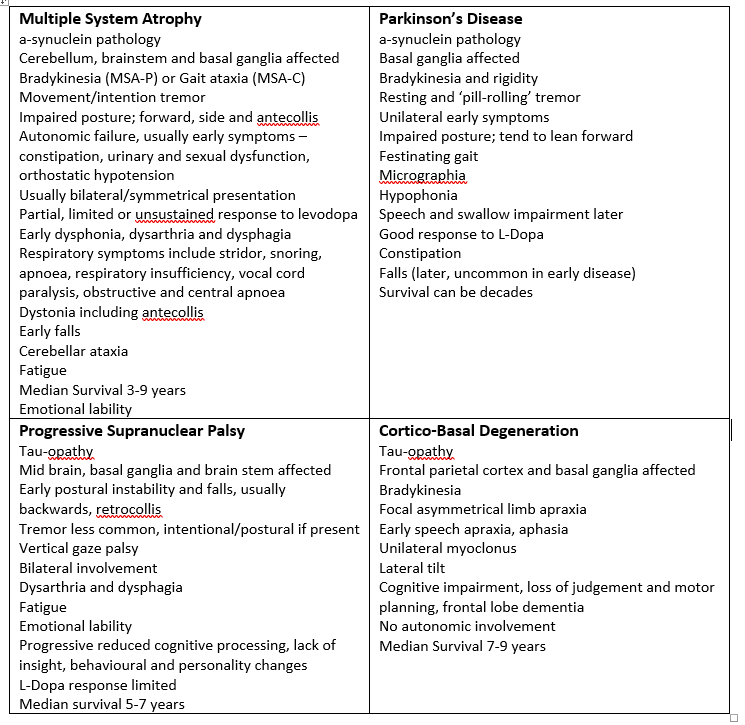
Distinguishing MSA from idiopathic Parkinson’s disease or cerebellar ataxia is still problematic, especially in the early stages. Speech and language therapists need to be aware of clinical features that characteristically distinguish symptoms of MSA from other Parkinsonism syndromes including PD, although these can be hard to discern in the early stages (see Table 1).
The key distinguishing clinical signs at diagnosis are [7]:
- Autonomic failure which includes orthostatic hypotension and bladder dysfunction (with erectile dysfunction in men)
- Poor response to levodopa (may receive partial or transient benefit)
- Akinetic rigid parkinsonism (present in 58% of cases) or cerebellar ataxia (29%)
MSA phenotypes
The criteria used most often to classify MSA recognise two main phenotypes [7]. In general, people present with predominance of Parkinsonian features (MSA-P) or predominance of cerebellar features (MSA-C), however symptoms overlap. All people with MSA develop autonomic symptoms.MSA is a rapidly progressing, neurodegenerative disorder leading to severe disability. It has been established that the prognosis is poorer in people who present with early autonomic dysfunction. People who have been classified as MSA-P are more likely to have greater functional decline [49]
MSA-P
The motor symptoms characteristic of MSA-P are similar to those observed in typical Parkinson’s disease and include rigidity, bradykinesia, tremor and poor balance. However, autonomic symptoms can also predate motor symptoms in MSA-P.
MSA-C
Cerebellar symptoms in MSA originate from the trunk spreading to the lower limbs which eventually affect gait. Gait ataxia, limb kinetic ataxia and dysarthria as well as cerebellar occulomotor disturbances are typical motor symptoms of this phenotype.
Treatment strategies
There is currently no consensus on the stages of disease progression in MSA, nor is it clearly defined. Average survival is close to a decade from symptom onset [8], although this is a guide only. Treatment varies for each stage of the disease and Speech and Language Therapists are urged to use their clinical reasoning skills based on the knowledge of the neuropathology of the disease. Patient centred goals should be realistic and appropriate, and multi-disciplinary intervention is key to the provision of a quality service. Medical management is based on symptom alleviation, most notably bradykinesia and orthostatic hypotension [40]. The table below summarises key medical interventions.
| Symptom | Intervention |
| Parkinsonism | L-Dopa (40-60% of MSA patients will initially respond) |
| Motor impairment | Physiotherapy and Occupational therapy |
| Orthostatic hypotension | Non-pharmacological, TEDS, abdominal binder, fluids, small meals, positioning and exercise advice. Pharmacological – fludrocortisone, midodrine. |
| Neurogenic urinary tract dysfunction | Non-pharmacological strategies, medications and catheterisation. Urology or continence team involvement. |
| Constipation | Exercise, diet, fluids and medications. Continence team involvement. ate |
| Erectile and sexual dysfunction | Specialist referral to andrology or sexual dysfunction clinic |
| Breathing problems | Physiotherapy, respiratory team, sleep studies, CPAP, tracheostomy |
| Dystonia/pain | Physiotherapy, positioning, support, massage, medications, Botox |
| Camptocormia | Physiotherapy and Occupational therapy support |
| REM Sleep disorder | Sleep studies, clonazepam, melatonin |
| Depression | Non-pharmacological, psychology and psychiatry support, pharmacological interventions |
| Speech and swallow problems | Speech and Language Therapy |
Table 2: Key symptoms and management. Adapted from Flabeau et al [9]
Speech and Language Therapy
Speech and Language Therapists (SLTs) play a vital role in assessing and providing therapeutic input for MSA patients with communication, swallowing and voice difficulties. Research surrounding the efficacy of speech and language therapy intervention with MSA is still limited and SLTs may only see a few clinical cases over their entire career, due to the rarity of the condition [6].
SLTs must apply a patient centred and holistic approach when working with this client group [7]. The differential diagnosis of MSA is still problematic and early symptoms can often be likened to those seen in patients with Parkinson’s disease. Both MSA and Parkinson’s disease present with progressive dysarthria, progressive dysphagia and reduced volume of voice. As a result of their similarities, SLTs could consider studies carried out with Parkinson’s disease or ataxic patients to help inform their early interventions.
Family members will often play a key role in supporting interventions, especially towards the advanced stages of the disease. They will often be responsible for supporting mealtimes and encouraging the person to use SLT techniques to aid communication during everyday life. Working with family members is critical to aid understanding of strategies for both swallowing and communication. Family members can be supported to adapt their own style of communication to support that of the person with MSA.
The benefits of managing patients with multi-disciplinary teams has been demonstrated across a number of conditions, and the approach particularly important when working with people who have progressive neurological conditions [56]. Liaison with specialist nurses, physiotherapy, occupational therapy, dietetics, neurology and their medical team will therefore be crucial to ensuring the person receives the right level of care.
People with MSA will often exhibit symptoms of impaired communication or swallowing difficulties much earlier than those with Parkinson’s disease [50]. The authors report dysarthria latencies for MSA and Progressive Supranuclear Palsy (PSP) as an average of 24 months post-diagnosis compared to 84 months for those with Parkinson’s [50]. Another study of five patients with MSA identified mild dysarthria symptoms as early as six months and severe impairment as early as one year after the onset of motor symptoms [14]. Earlier dysphagia symptoms are directly correlated with a generally shorter life expectancy [50]. Once communication, swallowing or voice difficulties emerge, they are also likely to decline more rapidly than in those who are diagnosed with Parkinson’s [51]. This can cause distress and requires the SLT to be able to take an informed approach to carrying out any interventions. Reviews may need to be more frequent and as such people with MSA may not be discharged from care depending on their local health authority’s policies and procedures. It is also important to ensure they know exactly how to contact their Speech and Language Therapist if things change. Voice and message banking should be considered as soon as possible after diagnosis to achieve optimum results before speech declines.
Communication Difficulties in MSA
The communication difficulties associated with MSA will differ from person to person and depending on the phenotype, although most patients will present with signs of hypokinetic, ataxia as well as spastic dysarthria [12, 13, 14, 15, 16, 57]. Rusz et al.’s [57] study of 40 people with MSA indicated that slow rate, inappropriate silences and irregular motion rates were features commonly found across both phenotypes. In addition, people with MSA-P mostly presented with imprecise articulation, harsh voice, monopitch, excessive pitch fluctuations and pitch breaks, whereas those with MSA-C were more prone to phoneme prolongations, audible inspiration and voice stoppages [57]. They also found that although the speech of people with MSA-P will often appear to be relatively similar to those with Parkinson’s they had more severe speech impairment, confirming the results of an earlier studies, e.g. [10, 24].
Common Features Impairing Communication in MSA
- Dysarthria; specifically hypokinetic dysarthria, ataxic dysarthria, spastic dysarthria or a mixture (mixed dysarthria)
- Dysprosody
- Dysphonia; may present with a hoarse or harsh voice or have reduced loudness
- Progressive limb weakness; impacting on the use of gesture
- Masked facial expression (orofacial dystonia); reducing non-verbal communication
- Decreased rate of speech
- Dysfluency
- Increasing cognitive difficulties, resulting in difficulty processing language to understand it and difficulties with verbal expression, which can affect ability to hold a conversation
- Plan for people with MSA to lose the ability to speak; consider voice and message banking and ensure forward planning regarding use of AAC
Assessing Communication Difficulties in MSA
The following aspects should be taken into consideration when assessing a person with MSA:
- Dysarthria – A dysarthria assessment should include auditory-perceptual clinical assessment and instrumental assessment if appropriate or available [17, 12]. A perceptual assessment may include the use of informal rating scales or formal tools such as the The Frenchay Dysarthria Assessment (FDA-2) [18]. Standardised reading passages such as ‘The Grandfather Passage’ may also be a useful resource to help collect auditory-perceptual information. Any findings from the perceptual assessment should help to inform whether instrumental assessment would be beneficial. In the clinical setting, it is unlikely that an instrumental assessment of the person’s dysarthria would drastically change the clinical management. It is important to consider the person’s speech in the context of their usual environment, in order to recommend appropriate strategies.
- Receptive Language – A person with MSA may find processing complex auditory or written information increasingly difficult, due to increasing cognitive difficulties [19, 20, 21]. Simple orientation tasks and assessing their ability to follow instructions of varying complexity should help to establish their current receptive capabilities [22]. Formal assessments of receptive language may be attempted but may not be appropriate depending on the stage of the MSA.
- Expressive Language –Is also affected due to the increasing cognitive impairments. A person with MSA experiences increasing difficulties with word finding and speech initiation, due to progressive neurological deterioration [23, 22].
- Dysphonia – A perceptual voice analysis tool such as the GRBAS [25] or CAPE-V [58] may be useful to record the impact of MSA on the patient’s vocal output. Pitch is often increased in people with MSA [24] there may be increased hoarseness and breathiness on phonation [57]. Due to the interconnectivity between dysarthria and dysphonia, the term dysarthrophonia can be used to describe the presentation.
- Inspiratory stridor – Defined as the presence of “turbulent airflow through a partially obstructed airway” [27], this symptom increasingly impacts on the MSA patient’s ability to breathe effectively [12]. Therefore, there will be an impact on their communication and connected speech output due to reduced respiratory support. The patient should seek medical support from ENT if inspiratory stridor is identified (ibid), as they may require surgery such as a tracheostomy procedure and it may cause a medical emergency if the airway is completely obstructed.
Potential Interventions to Support Communication
Voice banking, message banking and video banking
Voice and message banking should be discussed with the person with MSA as soon as possible after diagnosis. Local referral pathways should allow for SLT input for this specific task in the absence of any changes to speech, and the MDT should be encouraged to make these early referrals. This will allow for optimum recordings to be made before speech deteriorates,
Voice banking refers to the process of recording a set inventory of phrases, which are then used to create a computerised version of the person’s own voice for use on a voice output communication aid. The person can then generate an infinite number of novel utterances using their computerised voice. The MSA Trust will fund voice banking costs for all fully registered members in the UK and Ireland.
Please bear in mind that people will need to be able to use an aid in order to use their banked voice, and this my become more difficult in terms of cognition and/or mobility as MSA progresses. Speech and Language Therapists may need to manage their patients expectations around length and practicality of use.
Message banking allows a person to record their own choice of personal phrases for later use on a voice output communication aid. A computerised voice is not created with message banking, and therefore playback is limited to the phrase that has been recorded. However, it is the person’s exact voice that is played back rather than a computerised version and is therefore excellent for very personal phrases such as ‘I love you’.
Video banking refers to the process of video recording speech for a particular purpose, such as a special occasion in the future. This allows a person to capture additional of their communication, such as non-verbal aspects, and has a strong legacy work connection. Some people with MSA may also wish to consider this work.
See Benson (2022) in Walshe and Miller (2022) [73], chapter 8 for a case study using voice banking, message banking and video banking with a person with MSA.
Direct evidence-based research into appropriate communication interventions for people with MSA is an area that is significantly lacking in research. Given the presence of hypokinetic and ataxic dysarthria features, similar approaches to treatment could be effective. However, evidence for these disorders has also not been fully established, although more and more studies are emerging. For example, recent reviews on the effects of speech therapy on people with Parkinson’s [60, 61] indicate benefits on at least some speech outcome variables, particularly for loudness. Similarly, whilst there remains a lack of randomised controlled trials in the field of ataxic dysarthria [59], a number of smaller pilot studies have been published recently, either focusing on MSA [62, 63] or other types of progressive ataxias [64, 65, 66, 55], highlighting the potential of these approaches to improve communication.
SLTs should therefore take guidance from the wider literature focusing on other relevant disorders as well as management guidelines e.g. [28] to develop appropriate management plans for people with MSA. In doing so, they should bear in mind the stage of MSA. For advanced stage MSA, direct therapeutic input is unlikely to be beneficial due to the increasing cognitive impairment [19, 20] and advancing disease process. In relation to Alternative Augmentative Communication (AAC) use, AAC use should be considered and reviewed regularly throughout the disease progression. It may be that changes in cognition and face and eye movement mean that a high-tech system that was useful initially becomes less useful over time.
- AAC – following appropriate assessment. If AAC is deemed appropriate, the SLT should consider using both low technology and mid/high technology aids. A referral should be made to the regional assistive technology hub (or other service in your local area as necessary) covering the geographical area for assessment of AAC alongside EC (environmental controls) if the patient meets the criteria. AAC and EC systems should be considered together as a whole to ensure that the patient only needs to utilise one system for both. This referral can usually take place once the patient is having difficulty with direct access to more mainstream solutions (they are struggling to activate keys with their fingers). In some cases, the hubs will take referrals in advance of this if it is felt that the disease process is particularly rapid Oral motor exercises – although non-speech exercises remain a common feature in dysarthria therapy [67], there is currently no evidence that they have beneficial effects on speech outcomes [68].
- Compensatory dysarthria strategies – These strategies can be critical in ensuring that the person with MSA can communicate with their voice for as long as possible. Consider advising on reducing background noise, managing fatigue, pacing and SLOP strategies (Slow, Loud, Overemphasise, Pause) Identifying causes for conversational breakdown and relevant communication partner training should also be considered [33].
- Encouraging the use of deeper breathing techniques – such as diaphragmatic breathing [31] which can help increase subglottic pressure and increase volume [32].
- Communication support groups – Group approaches have found to be a beneficial way to implement therapy for people with a wide range of speech disorders and have demonstrated benefits both for communication as well as psychosocial wellbeing [55, 69].
- Vocal Exercises –A formal training programme such as Lee Silverman Voice Therapy LOUD programme (LSVT®) technique [34,35,77] or SPEAK OUT® may be beneficial in the earlier stages of the condition [62] although evidence is limited.
Examples of AAC Aids
Low Technology Aids: These may be text based or picture based, or a combination of both. Pictures and words together may be the best option when cognition is deteriorating.
- Simple alphabet charts – These can support a person with dysarthria, however various font sizes and colour combinations should be assessed as vision can become impaired.
- A personalised communication passport – This can gather relevant information about an individual, their wants, needs and preferences around a large number of areas. It can also offer some information about the condition. They are particularly useful if the person with MSA has to be admitted to hospital.
- Communication books – for members of the multi-disciplinary team and family to use with the patient. Taking time to ensure they contain up to date and relevant information makes them a good resource.
High Technology Aids:
- Tablet based computer devices can utilise a variety of specialised communication software, which may be text based, picture based or a combination of the two. Specialist assessment from an AAC hub can help with determining what is appropriate, and the SLT will need to review regularly to ensure the person with MSA can still use the system effectively.
- AAC can be accessed in a number of ways, including switches, eye gaze and head mouse. This will also be part of a specialist assessment and should be reviewed regularly.
- Light-writers – Can be helpful as a text to speech method that requires less confidence with technology. The person with MSA will need to be able to type onto the device, therefore if hand function deteriorates, they may no longer be useful. We can loan to members on a long-term loan basis.
- Amplifiers – to help support voice projection. We can loan these to members on a long-term loan basis.
- iPad / android tablet – There are a number of text to speech and picture to speech apps available on both apple and android devices. These may be particularly useful in the earlier stages of MSA if someone is struggling with their speech, but still well able to use everyday forms of technology.
Summary of the Key Communication Points:
- Voice and message banking should be discussed as early as possible
- People with MSA can experience an earlier onset of dysarthria compared to people diagnosed with Parkinson’s disease.
- The dysarthria is also likely to be more severe.
- There is a higher incidence of mixed dysarthrias in MSA, involving a combination of ataxic, spastic and hypokinetic features
- People with MSA will generally use fewer syllables per second in their speech due to the increased bradykinesia
- Intelligibility is likely to be reduced due to co-ordination difficulties of the lingual musculature and general muscle atrophy
- They may experience increased respiratory difficulties due to inspiratory stridor
- Cognitive decline in MSA may impact the use of AAC and overall communication ability.
Swallowing Difficulties in MSA
Research studies reflect that there is a high incidence of dysphagia occurring in people with MSA [37]. The oral stage of swallowing involves the person being able to masticate the bolus adequately and then propel the bolus posteriorly to the pharynx [38]. In a person with MSA, the propulsion of the bolus is likely to be delayed and extremely effortful, resulting in a significantly prolonged oral stage of the swallow [39]. This puts them at higher risk of aspiration as the reduced co-ordination may lead to a premature spillage of bolus. The SLT should also be aware of mealtimes difficulties that frequently occur in addition to dysphagia in patients with cognitive decline and consult the multi-disciplinary team on how best to manage these [71].
As with speech symptoms, the onset of dysphagia may differ from Parkinson’s and PSP. Dysphagia latencies are typically 67 months post diagnosis for people with MSA, compared to 42 months in PSP and 130 months in Parkinson’s disease [50]. Thus, people with PSP are more likely to experience dysphagia symptoms earlier than MSA patients, who in turn experience dysphagia significantly sooner than people with Parkinson’s disease.
Common Features Impairing Swallowing in MSA
As with other Parkinson plus diseases, people with MSA are likely to experience progressive oropharyngeal dysphagia. This can be caused by any of the following factors and a person may present with these symptoms:
- Coughing or choking on food or fluids due to reduced ability to protect the airway
- Increasing bradykinesia; making independent feeding increasingly difficult
- Poor hand to mouth co-ordination
- Decreased lip seal and anterior loss of saliva
- Fasciculation of the tongue; this can make manipulation of a bolus within the oral cavity very difficult. It can also impact the anterior to posterior propulsion of a bolus.
- Rigidity; impacting on the muscles required for adequate mastication
- Reduced appetite
- Impact of medication; if medication is not taken on time, the swallow function may deteriorate
A person with MSA-C is more likely to find the co-ordination of a bolus more difficult due to their ataxia.
Assessing Swallowing Difficulties in MSA
Any person who presents with a dysphagia should be thoroughly assessed in accordance with current RCSLT dysphagia clinical guidelines [8].
The SLT should consider:
- Carrying out a thorough cranial nerve examination –weakness in all of the cranial nerves involved in swallowing is likely
- Alertness and positioning of the patient prior to assessment; you may need to access your physiotherapy team to aid with positioning
- Previous levels of oral intake and any reported difficulties
- Any previous dysphagia recommendations that are in place
- Current respiratory status
- Carrying out a formal clinical bedside evaluation of swallow
- Referring for instrumental analysis of swallow as appropriate; however, a procedure such as video-fluoroscopy may not be appropriate in the advanced stagesAny advanced care planning which may be in place for the patient, when considering alternative feeding measure. It is critically important to consider quality of life and social aspects of eating and drinking in the context of dysphagia management for people with MSA. RCSLT guidance on eating and drinking with acknowledged risk should be used to support discussions [74].
It is good practice to discuss these decisions with the person with MSA early in their journey to understand their wishes whilst they have capacity to make their own decisions about eating and drinking at risk. The SLT should liaise closely with the MDT around all aspects of discussions and decisions around eating and drinking.
Mouth care
It has long been acknowledged that there is a link between poor oral hygiene, the presence of dysphagia and the subsequent risk of developing aspiration pneumonia [78]. It is therefore important for the treating clinician to consider providing education and training as required, to promote ongoing good oral hygiene for people with MSA. Mouth Care Matters is a campaign that was run by Health Education England and can provide you with further guidance and resources to share with clients and their families.
Potential Interventions to Support Swallowing
- Appropriate saliva management –Some people with MSA may struggle to control excess saliva and the SLT should consider this and seek medical input if felt appropriate.
- Modified diet consistencies –It may be appropriate to consider modifying consistencies of food and fluid as disease progresses. This may be part of a full swallowing and risk assessment. Recommendations should be in line with IDDSI guidelines [75].
- Swallow therapy exercises may be recommended by an SLT based on each individual’s needs – There are some small scale intensive dysphagia studies within the population of MSA however more research is needed at this current time [72].
- Carer training – (as with other progressive neurological conditions,) carers often play a vital role in ensuring safe delivery of nutrition and hydration by following SLT recommendations.
- Environmental adaptations – adaptive cutlery or plate guards to aid mealtimes and maintain independence and dignity for as long as possible. Devices such as a neater-eater – https://neater.co.uk can help some people with MSA. Smaller meals little and often or moving their main meal to lunch time may also help with fatigue. Joint working with MDT colleagues in Dietetics, Occupational Therapy and Physiotherapy is advised [76]
- Alternative methods of feeding – A nasogastric tube or a percutaneous endoscopic gastrostomy (PEG) to ensure adequate hydration and nutrition if appropriate (this should be discussed as part of an MDT). PEG for fluid intake may be a particularly relevant option if the person has symptoms of postural hypotension. Due to the progressive nature of the condition, the concept of alternative methods of feeding should be discussed during the early stages of MSA. It is important to inform patients that the presence of a tube for feeding support does not stop them from continuing to have food and fluid orally.
- Medication – If a patient is struggling with swallowing solids, they may need their medication altered to a more suitable form. The SLT could liaise with the patient’s GP or pharmacy directly.
Inspiratory Stridor
Inspiratory stridor is a symptom that is likely to emerge towards the latter stages of the condition [40]. This is likely to have a negative impact on the safety of the swallow, but also affect vocal output [41].
There is currently mixed research as to why this occurs in people with MSA. One theory is that it is caused by vocal fold adductor paralysis [42, 43]. Increased neurogenic atrophy within the intrinsic laryngeal muscles specifically, the posterior crico-aryteniod muscle, will also directly affect voicing ability [44]. Another theory is that poor vocal fold adduction is in fact caused by vocal fold dystonia [45]; which some studies suggest can be reduced by the use of Botox [46].
SLTs may notice that their patients with MSA will have more breathing difficulties and increased dysphonia. This will result in decreased vocal power and a significantly increased risk of aspiration during swallowing, and thus pneumonia [43]. The small apnoeic period that is experienced during swallowing may also be increased, causing greater fatigue during eating and drinking activities. The latter may result in reduced oral intake and it is therefore important to involve the dietician to manage this.
Speech and Language Respiratory Care Specialists are emerging within the clinical field. However, due to the significant lack of these clinical roles within clinical teams, the SLT should liaise with their respiratory team if the stridor becomes particularly intrusive on activities of daily living. Appropriate respiratory measures could then be applied if necessary.
Speech and Language Therapy in action: Case Example 1
Background
Patient X was a 66 year old lady diagnosed with probable MSA-P who was referred to her local Speech and Language Therapy service 12 months post-diagnosis. She was referred by her GP for increasing difficulties with her speech. X had reported no concerns regarding her eating and drinking ability, yet she was now avoiding high risk consistencies such as nuts and toast. It was also taking her longer to finish her meals and her husband supported her with this and other activities of daily living.
X was seen in an outpatient clinic for assessment, alongside her husband who was her main carer. X was predominantly wheelchair bound as the condition had significantly impacted on her mobility over the past 12 months. X was a smoker and had a pre-existing chronic obstructive pulmonary disease, which impacted on her breath support during conversation.
Assessment
An oral motor cranial nerve assessment was carried out with X to view any significant neurological impairment. The assessment revealed that she had reduced range of movements in her jaw (cranial nerve V), lips (cranial nerve VII), soft palate (cranial nerves IX & X) and tongue (cranial nerve XII). Although mild, these symptoms were negatively impacting on her ability to produce clear speech. Laryngeal movement was also significantly reduced on tactile assessment and her voice was markedly weak in presentation. The therapist therefore carried out a perceptual analysis of her voice using the GRBAS scale [25].
Her results are depicted as follows:
| Grade | Roughness | Breathiness | Asthenia | Strain |
| 2 | 2 | 1 | 1 | 1 |
It is not uncommon for voice and speech to also be particularly affected in MSA due to a decline in oro lingual strength [13, 10]. The literature suggests that in people with a Parkinsonian related disease, it is not uncommon for their palatal movement to be significantly reduced on phonation [47]. She was asked to read ‘The Grandfather Passage’, [48], which revealed how her intelligibility decreased with utterance length, as expected from the literature.
In a quiet one to one environment in a clinic, she was largely intelligible; however, this was likely to decrease significantly in a noisier environment.
Her dysarthria could be characterised as a mixed dysarthria with spastic and hypokinetic elements.
Management
X was encouraged to implement the following advice to help minimise the effects of her dysarthria. Her husband was also present to help implement the strategies:
- Avoid particularly loud or noisy environments
- Ensure people face you directly when talking with them
- Use shorter sentences or key words instead of long complex sentences
- Repeat key words if necessary or rephrase
- Spelling out words or using topic cues (or combination of both) can be useful to help people understand you
- Check that someone has thoroughly understood you
- Consideration was given towards using simple alphabet charts and speech apps, but was felt not to be appropriate at the time of assessment.
- A discussion also took place with X and her husband around voice and message banking, framed within the context of this being an ‘insurance policy’ for her voice in case she were to lose it further down the line and require AAC to communicate. X was interested in completing both voice and message banking, and a further appointment was made to commence this work with her as soon as possible.
Outcome
X was still a functional and competent communicator who only required minimal environmental considerations and prompts for clearer articulation. At the time of assessment, X was still able to converse on the telephone. However, if she had been struggling with this she may have been advised to consider using a pre-prepared phrase in order for unfamiliar listeners to give her more time to speak. For example ‘I have a difficulty with my speech, please be patient’. This could be facilitated by her husband and other family and friends.
X was provided with recommendations to take away and they were advised that if they felt they had deteriorated further, to contact the speech and language therapy department. A review appointment was suggested in 6 months, but X advised she could contact the SLT department for a review sooner if she fet her speech had changed.
Reflection
One point that X and her husband both commented on was the rapid deterioration of mobility and general motor function (which in this case was impacting her speech). Over just 12 months, X had gone from being completely independent to requiring support for most activities of daily living.
The emotional impact of this was important for the SLTto be able to empathise with and having prior knowledge about the rate of deterioration helped support the session and patient.
This may also reinforce the benefit of early discussions about future communication needs depending upon how much the person with MSA knows and understands about the course of their condition.
Case Example 2
Referral
A patient has been referred to the Speech and Language Therapy department following concerns relating to the safety of their swallow. The patient has a known probable diagnosis of MSA and is in the latter stages of the condition. The patient is 80 years old and has a supportive family. The medical team have requested a review is carried out whilst he is an inpatient to carry out an assessment and provide recommendations.
Dysphagia Assessment
Prior to assessment consent should be obtained from the patient, if the patient is felt to lack capacity to do this an assessment may be carried out in the patient’s best interests.
In line with clinical dysphagia guidelines set out by the RCSLT, an oro-motor assessment may be carried out in order to observe the impact of muscle weakness on the swallowing mechanism. It is likely that cranial nerves V, VII, X, XI and XII will all be directly affected and therefore reduce the safety and co-ordination of the swallow.
A clinical evaluation of swallow may then be carried out with the patient if appropriate. This can involve trialling modified consistency diets and establishing the safest form of oral intake for the patient. If the patient is thought to be acutely unwell, it may be appropriate for the medical team to consider alternative forms of nutrition and hydration, such as a nasogastric tube (NGT). However, it would also be important to consider their current respiratory status, due to the known co-morbidity of inspiratory stridor in some patients with MSA.
Dysphagia Management
Swallow therapy exercises such as a chin tuck or supraglottic swallow could reduce the risk of aspiration [38, 7] and improve co-ordination.
However, it is unlikely that a patient would be able to retain and use these strategies effectively at every mealtime if they were in the latter stages of MSA. Instead, a modified consistency diet may be considered (if available) with support and assistance from carers. Consideration towards appropriate saliva management should also be discussed if the patient was not already receiving treatment.
Other Considerations
Ataxic symptoms and problems with overall co-ordination can be a particular problem for patients with probable MSA [49]. It can result in an uncoordinated swallow, especially during the oral stage [39]. MSA-P patients can experience on/off periods with medication; the impact of which is likely to affect a patient’s ability to communicate, swallow and mobilise [2].
As people with MSA often require regular medication [51], if they are seen to be struggling with solid tablet dose medications, an alternative form should be considered e.g. liquid or dispersible.
The SLT can therefore advise the patient’s GP or medical team to liaise with a pharmacist to identify the appropriate alternatives available.
Summary
At present the exact nature of how quickly a swallow may deteriorate in a person with MSA is unclear. Recommendations may therefore be set with the knowledge that Speech and Language Therapy can be contacted if a review is required. For most patients with MSA, it is unlikely that a referral onto an instrumental dysphagia assessment (such as video fluoroscopy) would change the dysphagia management.
If X’s swallow was seen to deteriorate further and they were aspirating on a range of consistencies, alternative feeding may be considered dependent on the individual [8]. This is the use of either a nasogastric tube (NGT; acute periods) or a percutaneous endoscopic gastrostomy (PEG; chronic periods) in order to maintain nutrition and hydration needs. Some researchers support the use of PEG insertion in MSA patients with severe dysphagia. They suggest that if a person with MSA suffers from severe dysphagia, a PEG may help reduce the risk of aspiration, provide adequate calorie intake and ensure medications are being received [52, 7, 51].
However, Gazulla et al [53] stated that although a PEG may reduce the risk of aspiration on food and fluids, a patient with MSA may still potentially aspirate on their own saliva.]. It may only prolong a poor quality of life, so these factors would need to be thoroughly discussed with the patient, the family and medical team. Due to the poor prognosis for patients with MSA, alternative feeding may not be considered appropriate and a decision to “feed at risk” may be made by the medical team. Continue eating and drinking with acknowledged risk may be the most suitable option, in liaison with the medical team and wider MDT.
If it was decided that a patient was not appropriate for a PEG placement, an appropriate management plan for the family should be put in place. A report from Speech and Language Therapy may also be required in order to support appropriate care funding if required.
What next?
With the exception of one study [72], there are no direct studies assessing appropriate therapeutic interventions for dysphagia in MSA [7]. However, parallels may be drawn between MSA and Parkinson’s disease dysphagia management strategies due to the similar nature of their difficulties. Attention to MSA-specific characteristics such as laryngeal stridor and ataxia will be important for the SLT to consider in relation to future dysphagia management.
Written by Lauren Gray, Speech and Language Therapist with special thanks to support from Gloucestershire Care Services Speech and Language Therapy department and members of the RCSLT. Updated by Speech and Language Therapists and Researchers at review in February 2025.
References
- A. Schrag, Y Ben-Shlomo, and NP Quinn. Prevalence of Progressive Supranuclear Palsy and Multiple System Atrophy: a cross sectional study. The Lancet. 2006; 354: 1771-1775.
- Parkinson’s UK website https://www.parkinsons.org.uk/information-and-support/
- Haw C, Trewhitt T, Boddy E, Evans J. Involving carers in communication groups for people with Parkinson’s disease. (1999). Parkinson’s Disease: Studies in Psychological and Social Care. BPS Books, Leicester, UK: Percival R, Hobson P (Eds).
- PA Hanna, JJ Jankovic and JB Kirkpatrick. Multiple System Atrophy: The putative causative role of environmental toxins. Arch Neuro 199; 56:90-94.
- Swan L, Dupont J. Multiple system atrophy. Phys Ther. 1999;79:488–494.
- Schrag, A., Y. Ben-Shlomo, and N. P. Quinn. “Prevalence of progressive supranuclear palsy and multiple system atrophy: a cross-sectional study.” The Lancet 354.9192 (1999): 1771-1775.
- Wenning, G., Fanciulli, A. (2014). Multiple System Atrophy. Austria: Springer-Verlag Wien.
- RCSLT Clinical Information: Dysphagia / Eating, drinking and swallowing needs. https://www.rcslt.org/speech-and-language-therapy/clinical-information/dysphagia/. Webpage accesse 15/12/2022.
- Flabeau, O, Meissner, G and Tison, F (2010). Multiple System Atrophy: current and future approaches to management. Therapeutic Advances in Neurological Disorders. 2010;3(4): 249-263.
- Penner, H., Miller, N., & Wolters, M. (2007). Motor speech disorders in three parkinsonian syndromes: a comparative study. In J. Trouvain & W.J. Barry (eds), Book of Abstracts, 16th International Congress of Phonetic Sciences (ICPhS), 6-10 August 2001, Saarbrücken, Germany.
- Dronkers, N.F. (1996). A new brain region for co-ordinating speech articulation. Nature, 384, 159.
- Duffy, J. R. (2019). Motor speech disorders: Substrates, differential diagnosis, and management. 4th ed. Elevier, Mosby.
- Kluin, K.J., Gilman, S., Lohman, M., Junck, L. (1996). Characteristics of the dysarthria of multiple system atrophy. Archives of Neurology 53, 545-548.
- Knopp, D. B., Barsottini, O. G. P., & Ferraz, H. B. (2002). Multiple system atrophy speech assessment: study of five cases. Arquivos de neuro-psiquiatria, 60(3A), 619-623.
- Hartelius, L., Gustavsson, H., Astrand, M., Holmberg, B. (2006). Perceptual analysis of speech in multiple system atrophy and progressive supranuclear palsy. JMSLP 14, 241-247.
- Rusz, J., Bonnet, C., Klempíř, J., Tykalová, T., Baborová, E., Novotný, M., Rulseh, A. and Růžička, E. (2015). Speech disorders reflect differing pathophysiology in Parkinson’s disease, progressive supranuclear palsy and multiple system atrophy. Journal of neurology, 262(4), pp.992-1001.
- Murdoch, B. E. (1998). Dysarthria: A physiological approach to assessment and treatment. Nelson Thornes.
- Enderby, P., & Palmer, R. (2008). Frenchay Dysarthria Assessment – Second Edition (FDA-2). Austin, TX: PRO-ED.
- 1. Kawai, Y., et al., Cognitive impairments in multiple system atrophy. MSA-C vs MSA-P, 2008. 70(16 Part 2): p. 1390-1396.
- 1. Stankovic, I., et al., Cognitive impairment in multiple system atrophy: a position statement by the Neuropsychology Task Force of the MDS Multiple System Atrophy (MODIMSA) study group. Mov Disord, 2014. 29(7): p. 857-67.
- Spaccavento, S., Del Prete, M., Loverre, A., Craca, A., & Nardulli, R. (2013). Multiple system atrophy with early cognitive deficits: A case report. Neurocase, 19(6), 613-622.
- Apostolova, L.G., et al., Multiple system atrophy presenting with language impairment. Neurology, 2006. 67(4): p. 726-727.
- Cuoco, S., et al., The language profile in multiple system atrophy: an exploratory study. Journal of Neural Transmission, 2021. 128(8): p. 1195-1203.
- Huh, Y. E., Park, J., Suh, M. K., Lee, S. E., Kim, J., Jeong, Y., … & Cho, J. W. (2015). Differences in early speech patterns between Parkinson variant of multiple system atrophy and Parkinson’s disease. Brain and language, 147, 14-20.
- Hirano, M. (1981). Clinical examination of voice (Vol. 5). Springer.
- Haydock, S., Whitehead, D., & Fritz, Z. (Eds.). (2014). Acute Medicine. Cambridge University Press.
- Kalf JG, de Swart BJM, Bonnier M, Hofman M, Kanters J, Kocken J, Miltenburg M, Bloem BR, Munneke M. Guidelines for speech-language therapy in Parkinson’s disease. Nijmegen, The Netherlands / Miami (FL), U.S.A.: ParkinsonNet/NPF. https://www.parkinsonnet.nl/app/uploads/sites/3/2019/11/dutch_slp_guidelines-final.pdf. Webpage access 15/12/2022
- Fawcus, M., 2013. Voice disorders and their management. London: Springer.
- Bloch, S. and S. Beeke, (2021) A better conversations approach for people living with dysarthria, in M. Walshe and N. Miller, Editors,Clinical Cases in Dysarthria. Routledge: London.
- Ramig, L.O., et al., Intensive speech treatment for patients with Parkinson’s disease: short-and long-term comparison of two techniques. Neurology, 1996. 47(6): p. 1496-504.
- Ramig, L., et al., Speech treatment in Parkinson’s disease: Randomized controlled trial (RCT). Movement Disorders, 2018. 33(11): p. 1777-1791.
- Wenning, G. K., Ben-Shlomo, Y., Hughes, A., Daniel, S. E., Lees, A., & Quinn, N. P. (2000). What clinical features are most useful to distinguish definite multiple system atrophy from Parkinson’s disease?. Journal of Neurology, Neurosurgery & Psychiatry, 68(4), 434-440.
- Vogel, A.P., Fendel, L., Paige Brubacher, K., Chan, V. and Maule, R., 2015. Dysphagia in spinocerebellar ataxia and multiple system atrophy-cerebellar. Speech, Language and Hearing, 18(1), pp.39-43.
- Logemann, J.A. (1998). Evaluation and Treatment of Swallowing Disorders. Second Edition. Austin: PRO-ED Inc.
- Fernagut, P. O., Vital, A., Canron, M. H., Tison, F., & Meissner, W. G. (2012). Ambiguous mechanisms of dysphagia in multiple system atrophy. Brain;135(Pt 2):e205; author reply e206. doi: 10.1093/brain/awr185 .
- Flabeau, O., Meissner, W. G., Ozier, A., Berger, P., Tison, F., & Fernagut, P. O. (2014). Breathing variability and brainstem serotonergic loss in a genetic model of multiple system atrophy. Movement Disorders, 29(3), 388-395.
- Lalich, I. J., Ekbom, D. C., Starkman, S. J., Orbelo, D. M., & Morgenthaler, T. I. (2014). Vocal fold motion impairment in multiple system atrophy. The Laryngoscope, 124(3), 730-735.
- Isozaki, E., Shimizu, T., Takamoto, K., Horiguchi, S., Hayashida, T., Oda, M., & Tanabe, H. (1995). Vocal cord abductor paralysis (VCAP) in Parkinson’s disease: difference from VCAP in multiple system atrophy. Journal of the neurological sciences, 130(2), 197-202.
- Kurisaki, H. (1999). Prognosis of multiple system atrophy–survival time with or without tracheostomy. Rinsho shinkeigaku, Clinical neurology, 39(5), 503-507.
- Yokoji, I., Nakamura, S., & Ikeda, T. (1997). [A case of progressive supranuclear palsy associated with bilateral vocal cord abductor paralysis]. Rinsho shinkeigaku, Clinical neurology, 37(6), 523-525.
- Vetrugno, R., Liguori, R., Cortelli, P., Plazzi, G., Vicini, C., Campanini, A., & Montagna, P. (2007). Sleep‐related stridor due to dystonic vocal cord motion and neurogenic tachypnea/tachycardia in multiple system atrophy. Movement disorders, 22(5), 673-678.
- Merlo, I. M., Occhini, A., Pacchetti, C., & Alfonsi, E. (2002). Not paralysis, but dystonia causes stridor in multiple system atrophy. Neurology, 58(4), 649-652.
- Love, R.J., Webb, W.G. (2001). Neurology for the Speech-Language Pathologist (4th Ed). Butterworth: Heinemann.
- Darley, F. L., Aronson, A. E., & Brown, J. R. (1975). Motor speech disorders (3rd ed.). Philadelphia, PA: W.B. Saunders Company.
- Wenning, G. K., Geser, F., Krismer, F., Seppi, K., Duerr, S., Boesch, S., … & European Multiple System Atrophy Study Group. (2013). The natural history of multiple system atrophy: a prospective European cohort study. The Lancet Neurology, 12(3), 264-274.
- Müller, J., Wenning, G. K., Verny, M., McKee, A., Chaudhuri, K. R., Jellinger, K.,& Litvan, I. (2001). Progression of dysarthria and dysphagia in postmortem-confirmed parkinsonian disorders. Archives of neurology, 58(2), 259-264.
- Fanciulli, A., Wenning, G.K. (2015). Multiple system atrophy. New England Journal of Medicine, 372, 249-63.
- Mehanna, R., & Jankovic, J. (2010). Respiratory problems in neurologic movement disorders. Parkinsonism & related disorders, 16(10), 628-638
- Gazulla, J., Berciano, J., Gilman, S., Wenning, G. K., & Low, P. A. (2015). Multiple-system atrophy. The New England journal of medicine, 372(14), 1374-1374.
- Wenning GK, Stankovic I, Vignatelli L, Fanciulli A, Calandra-Buonaura G, Seppi K, Palma JA, Meissner WG, Krismer F, Berg D, Cortelli P, Freeman R, Halliday G, Höglinger G, Lang A, Ling H, Litvan I, Low P, Miki Y, Panicker J, Pellecchia MT, Quinn N, Sakakibara R, Stamelou M, Tolosa E, Tsuji S, Warner T, Poewe W, Kaufmann H. The Movement Disorder Society Criteria for the Diagnosis of Multiple System Atrophy. Mov Disord. 2022 Jun;37(6):1131-1148. doi: 10.1002/mds.29005. Epub 2022 Apr 21. PMID: 35445419; PMCID: PMC9321158.
- Lowit, A., Cox, J., Loucas, M., Grassly, J., Egan, A., van Brenk, F., & Hadjivassilliou, M. (2022). ClearSpeechTogether: a rater blinded, single, controlled feasibility study of speech intervention for speakers with progressive ataxia. The Cerebellum. https://doi.org/10.1007/s12311-022-01462-9
- Ward, C.D., et al., Multidisciplinary approaches in progressive neurological disease: can we do better? Journal of Neurology, Neurosurgery & Psychiatry, 2003. 74(suppl 4): p. iv8-iv12.
- Rusz, J., et al., Distinctive speech signature in cerebellar and parkinsonian subtypes of multiple system atrophy. Journal of Neurology, 2019. 266(6): p. 1394-1404.
- Kempster, G.B., et al., Consensus auditory-perceptual evaluation of voice: development of a standardized clinical protocol. Am J Speech Lang Pathol, 2009. 18(2): p. 124-32.
- Vogel, A.P., J. Folker, and M.L. Poole, Treatment for speech disorder in Friedreich ataxia and other hereditary ataxia syndromes. Cochrane Database of Systematic Reviews, 2014(10).
- Yuan, F., et al., Lee Silverman Voice Treatment for dysarthria in patients with Parkinson’s disease: a systematic review and meta-analysis. European Journal of Neurology, 2020. 27(10): p. 1957-1970.
- Muñoz-Vigueras, N., et al., Speech and language therapy treatment on hypokinetic dysarthria in Parkinson disease: Systematic review and meta-analysis. Clinical Rehabilitation, 2021. 35(5): p. 639-655.
- Park, Y., Efficacy of intensive treatment of dysarthria for people with multiple system atrophy. Phonetics and Speech Sciences, 2018. 10(4): p. 163-171.
- Sonoda, Y., et al., Short-Term Effect of Intensive Speech Therapy on Dysarthria in Patients With Sporadic Spinocerebellar Degeneration. Journal of Speech, Language, and Hearing Research, 2021. 64(3): p. 725-733.
- Vogel, A.P., et al., Speech treatment improves dysarthria in multisystemic ataxia: a rater-blinded, controlled pilot-study in ARSACS. J Neurol, 2019. 266(5): p. 1260-1266.
- Lowit, A., A. Egan, and M. Hadjivassiliou, Feasibility and Acceptability of Lee Silverman Voice Treatment in Progressive Ataxias. The Cerebellum, 2020. 19(5): p. 701-714.
- Vogel, A.P., et al., Home-based biofeedback speech treatment improves dysarthria in repeat-expansion SCAs. Annals of Clinical and Translational Neurology, 2022. https://doi.org/10.1002/acn3.51613
- Mackenzie, C., M. Muir, and C. Allen, Non-speech oro-motor exercise use in acquired dysarthria management: regimes and rationales. International Journal of Language & Communication Disorders, 2010. 45(6): p. 617-629.
- Mackenzie, C., et al., Non-speech oro-motor exercises in post-stroke dysarthria intervention: a randomized feasibility trial. International Journal of Language & Communication Disorders, 2014. 49(5): p. 602-617.
- Whillans, C., et al., A systematic review of group intervention for acquired dysarthria in adults. Disability and Rehabilitation, 2020: p. 1-17.
- Parkinson Voice Project: SPEAK OUT!® https://www.parkinsonvoiceproject.org, accessed 15/12/2022.
- Egan, A., C. Andrews, and A. Lowit, Dysphagia and mealtime difficulties in dementia: Speech and language therapists’ practices and perspectives. International Journal of Language & Communication Disorders. 55(5):777-792. doi: 10.1111/1460-6984.12563.
- Park, A., et al., Swallowing Outcomes Following Voice Therapy in Multiple System Atrophy with Dysphagia: Comparison of Treatment Efficacy with Parkinson’s Disease. Dysphagia, 2021. 37(1):198-206. doi: 10.1007/s00455-021-10265-9.73. Reference: Clinical Cases in Dysarthria (edited by Margaret Walshe and Nick Miller, 2022 – Routledge) Chapter 8: Saving lost voices: a toolkit for preserving communicative identity.
- https://www.rcslt.org/members/clinical-guidance/eating-and-drinking-with-acknowledged-risks-risk-feeding/
- www.iddsi.org
- Lindop, F and Skelly, R (eds), 2022, Parkison’s Disease A multidisciplinary Guide to Management. GB:Elsevier
- Hayashi K, Izumi R, Saito N, et al., Effectiveness of Lee Silverman Voice Treatment (LSVT-LOUD) in Parkinsonian-Type Multiple System Atrophy (MSA-P): A Case Report. Cureus 16(11): e73106, 2024. doi:10.7759/cureus.73106
- Ashford JR. Impaired oral health: a required companion of bacterial aspiration pneumonia. Front Rehabil Sci. 2024 Jun 4;5:1337920. doi: 10.3389/fresc.2024.1337920. PMID: 38894716; PMCID: PMC11183832.