This document serves as a guide to physiotherapists working with patients with multiple system atrophy (MSA). It draws on available literature in MSA, Parkinson’s disease and other atypical Parkinsonism disorders. It does not cover aetiology, epidemiology, neuropathology and medical management in any depth. Further reading on these topics and others can be accessed via the list of resources (including the MSA website), given at the end of the document.
The Multiple System Atrophy Trust (MSA Trust) produces a series of specialist MSA factsheets for health professionals to enable them to improve the treatment people with MSA receive. Other factsheets can be found on our website: www.msatrust.org.uk.
The Multiple System Atrophy Trust (MSA Trust) is the UK’s main charity supporting people with MSA. As well as helping people who have MSA, the Trust supports anyone affected by the disease, including carers, families, friends and health professionals.
The Trust employs MSA Health Care Specialists, manages a telephone and email advice service and runs a UK-wide network of support groups. It provides up-to-date literature for people affected by MSA and for health professionals. It also funds vital research to find the cause, and one day, cure for MSA.
To ensure services are accessible to everyone, the Trust is committed to providing its services free of charge. The MSA Trust is a charity funded entirely on voluntary donations.
The MSA Trust is always keen to receive feedback about the information it produces, please email support@msatrust.org.uk with any comments.
Introduction
MSA is a rare progressive neurological disorder that affects adult men and women and leads to premature death. Currently, there is no known cause or cure. MSA causes degeneration or atrophy of nerve cells in several (or multiple) areas of the brain which results in problems with movement, balance and automatic functions of the body such as swallowing, bowel, bladder and blood pressure control.
Globally, around 4.4 people per 100,000 have MSA which equates to almost 3,300 people living with MSA in the UK [1]. Parkinson’s disease is more common, affecting about 210 per 100,000 in the UK [2].
MSA usually starts between the ages of 50-60 years, but it can affect people younger and older. It affects both sexes [3].
Neuropathology
MSA is an alphasynucelinopathy, presenting pathologically with glial cytoplasmic inclusions and neuronal loss. The mechanisms underlying the condition and the factors that trigger MSA onset are yet to be established. Environmental and dietary influences have been cited [4] however definitive cause and risk factors are yet to be established. Symptoms of MSA are a manifestation of various pathologies originating in the striatonigral, olivopontocerebellar and central autonomic degeneration. This causes a combination of symptoms of Parkinsonism, Cerebellar signs and autonomic dysfunction.
![Figure 1 - MSA signs and symptoms (Adapted from Swan [5])](https://www.msatrust.org.uk/wp-content/smush-webp/2026/03/msa-signs-and-symptoms.png.webp)
Clinical diagnosis of MSA
The term MSA was first used in 1969 – prior to this it had previously been known as Shy -Drager Syndrome. The first diagnostic criteria for MSA were proposed in 1989 [6] and Second Consensus Criteria were defined in 2008 [7] which defined three levels of certainty of the diagnosis—possible, probable and definite MSA. New Movement Disorder Society(MDS) criteria for MSA diagnosis were published in 2022 using an evidence-based and consensus-based methodology[24]. Neuropathologically established MSA criteria remains the same, but new categories of clinically established MSA, clinically probable MSA and possible prodromal MSA are proposed.
Diagnosis of MSA is a clinical one and as such distinguishing MSA from idiopathic Parkinson’s disease is still problematic, with both presenting with abnormal DAT scans. Physiotherapists need to be aware of clinical features that characteristically distinguish symptoms of MSA from other Parkinsonism syndromes including PD, although these can be hard to discern in the early stages (see Table 1).
The key distinguishing clinical signs at diagnosis are [3]:
- Autonomic failure which includes postural hypotension, bladder and bowel dysfunction, sexual dysfuncion and possible speech and swallow impairment.
- Poor response to levodopa (may receive partial or transient benefit)
- Akinetic rigid parkinsonism (present in 58% of cases) or cerebellar ataxia (29%)
Table 1: Common and distinguishing features for PD, MSA, PSP and CBD (Courtesy of Katie Rigg, MSA Healthcare Specialist, Multiple System Atrophy Trust)
MSA phenotypes
The criteria used most often to classify MSA recognise two main phenotypes [3]. In general, patients present with predominance of Parkinsonian features (MSA-P) or predominance of cerebellar features (MSA-C), however symptoms overlap. Almost all patients with MSA develop autonomic symptoms preceded by motor symptoms. In Western hemisphere cohorts approximately 80% of cases are predominantly MSA-P. The contrary is true for Eastern hemisphere populations. This may be due to be to racial genetic differences and cultural characteristics. MSA is a rapidly progressing, neurodegenerative disorder. It has been established that the prognosis is poorer in patients who present with early autonomic dysfunction. Patients who have been classified as MSA-P are more likely to have greater functional decline [8].
MSA-P
The motor symptoms characteristic of MSA-P are similar to those observed in typical Parkinson’s disease and include rigidity, bradykinesia, tremor and poor balance. However, autonomic symptoms can also predate motor symptoms in MSA-P.
MSA-C
Cerebellar symptoms in MSA originate from the trunk spreading to the lower limbs which eventually affect gait. Gait ataxia, limb kinetic ataxia and scanning dysarthria as well as cerebellar occulomotor disturbances are typical motor symptoms of this phenotype.
Treatment strategies
There is currently no consensus on the stages of disease progression in MSA, nor is it clearly defined. Average survival is close to a decade [8], although this is a guide only. Treatment varies for each stage of the disease and physiotherapists are urged to use their clinical reasoning skills based on the knowledge of the neuropathology of the disease. Patient centred goals should be realistic and appropriate, and multi-disciplinary intervention is key to the provision of a quality service. Medical management is based on symptom management [9]. The table below summarises key interventions.
| Symptom | Intervention |
| Parkinsonism | L-Dopa (40-60% of MSA patients will initially respond) |
| Motor Symptoms | Physiotherapy and Occupational therapy |
| Orthostatic hypotension | Non-pharmacological, TEDS, abdominal binder, fluids, small meals, positioning and exercise advice. Pharmacological – fludrocortisone, midodrine, droxidopa |
| Neurogenic urinary tract dysfunction | Non pharmacological strategies, medications and catheterisation. Urology or continence team involvement. |
| Constipation | Exercise, diet, fluids and medications. Continence team involvement. |
| Erectile dysfunction | Specialist referral to andrology or sexual dysfunction clinic |
| Breathing problems | Physiotherapy, respiratory team, sleep studies, CPAP, tracheostomy |
| Dystonia/pain | Physiotherapy, positioning, support, massage, medications, Botox |
| Camptocormia | Physiotherapy and Occupational therapy support |
| REM Sleep disorder | Sleep studies, clonazepam, melatonin |
| Depression | Non-pharmacological, psychology and psychiatry support, pharmacological interventions |
| Speech and swallow difficulties | Speech and Language Therapy |
Table 2: Key symptoms and medical management Adapted from: Flabeau et al [9] and My Treatment Approach to Multiple System Atrophy – Mayo Clinic Proceedings [25] Coon and Ahlskog, 2020.
Physiotherapy
Evidence for the effectiveness of physiotherapy in MSA is restricted to an inter-disciplinary, inpatient intervention for a mixed diagnostic cohort [10], and a case-control study [11]. One UK study reported a positive effect of occupational therapy in MSA [12]. However, there isa significant body of research for physiotherapy for Parkinson’s disease which can be applied to MSA-P (see below). Physiotherapy evidence for MSA-C (and other degenerative ataxias) is limited, and there are no guidelines.
It is important to remember that MSA is progressive and maintenance (let alone improvement) is not always an indicator of effective intervention. Therapy must be integrated into daily living, and realistic programmes developed. Symptoms can vary on a daily basis and it is important to optimise exercise and activity during better days. Long, drawn out sessions of therapy input are unlikely to be successful [13]. A multidisciplinary approach is essential to MSA, and close liaison with the team is critical. Anticipatory planning is essential and integration of other team members’ input (e.g. speech and language therapy, wheelchair services, palliative care). Deterioration can be rapid especially as disease severity increases.
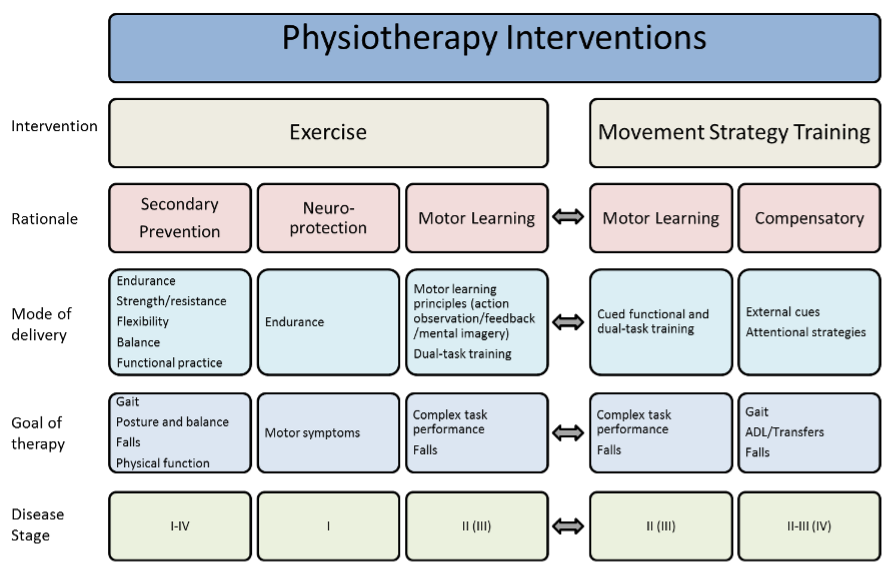
Fig. 2 broadly describes goals of physiotherapy over the time course of the disease.
We first describe physiotherapy intervention common to both phenotypes across a continuum of mobility, which may or may not correspond with time since onset. MSA symptoms may well be established at time of diagnosis, in which case at least some features of mobility will be impaired. Ideally, physiotherapy will begin prior to the presence of motor disturbance, and as soon as possible after diagnosis.
MSA is a rapidly progressive and aggressive disorder; planning and team effort is of paramount importance in management. The presence of orthostatic hypotension coupled with fatigue can make physiotherapy intervention challenging. However, fatigue should not stop patients from undertaking and benefiting from exercise. Graduated programmes of activity can improve levels of fatigue over time, but a balance must be achieved.
Physiotherapists also need to be aware of the importance of minimising the impact of the autonomic dysfunction on physical function. The following advice may be useful:
- If necessary, elevate the head of the bed (30°) during sleep – ideally use a mattress variator to achieve this
- Use TED stockings and abdominal binders to help symptoms of postural hypotension
- Ensure postural transitions are slow and controlled, especially on sit to stand when there is the greatest risk of a fall in blood pressure levels
- Avoid prolonged standing and/or activities that require prolonged stand
- Increase fluid intake especially before exercise
- Swimming or any exercise in a pool may be particularly beneficial because of the counteractive effect of hydrostatic pressure on postural hypotension, be careful of drop in blood pressure wehnexiting the water
- Be aware of the effect of warm environments and activities that may elicit the Valsalva manoeuvre (forcible exhalation whilst keeping nose and mouth closed)
- Stimulate BP regulation by, e.g. calf pump exercises prior to movement, crossing arms and legs, encouraging flexed postures
- Avoid exercise in the early morning and after large meals and ensure exercise is not carried out in a room that is too hot
- See MSA Trust factsheet on Postural hypotension

Preserved mobility
It will be important to establish a relationship with the person with MSA and their family who may also wish to encourage and be involved in physical activity and physiotherapy. Gently assist with coming to terms with diagnosis, if appropriate.
General advice may be all that is required if motor control is essentially preserved. Formal exercise should be encouraged (group classes, daily routines, gym attendance), but for people who have never been interested in this kind of exercise, a broader definition is required. It will be more useful to talk about maintenance of ‘activity’ (gardening, walking the dog, walking to the shops) rather than ‘exercise’.
Detailed assessment will reveal any early, subtle changes in performance, e.g. use of a broader base of support to maintain postural control and gait, inability to respond to unexpected perturbations, abnormal synergistic patterns of movement. In such cases, physiotherapy needs to be selective and appropriate to ameliorate motor impairment. Focus on postural control and identify triggers for falls. The strongest predictor of future falls is a previous fall, by which time associated risk factors such as fear of falling may also be evident. Physiotherapists need to identify falls risk earlier than this, and pre-empt the initial fall. This is challenging, but comprehensive assessment of postural control and falls risk during this stage is critical.
Stress the system in clinic (or home) by testing gait under dual task conditions (gait plus concurrent cognitive task) and increase environmental difficulty (over obstacles, through narrow walkways etc.). Examine postural control strategies using assessment tools that reveal underlying deficit (e.g. the BESTest) [14], rather than relying solely on functional tasks. Use attentional strategies to compensate for motor deficit and produce effective movement. This is especially critical if PD signs
predominate. Use of outcome measures may be relevant (e.g. the Unified MSA Rating Scale (UMSARS) or timed walks) if monitoring change is important.
Respiratory symptoms such as sleep apnoea, dysrhythmia and stridor can manifest early on [9] and specific assessment warranted. Check breathing techniques and if appropriate, link techniques to exercise and activity at this stage.
Impaired mobility
As motor and autonomic symptoms progress, selective input is required to optimise performance. General advice regarding exercise and/or activity may be less relevant. Focal dystonia may be evident and require symptomatic relief such as botulinum toxin therapy. Physiotherapy for focal dystonia may be worth considering (e.g. gentle stretching, support, sensory input), although there is no quality evidence to support efficacy. When assessing gait and functional mobility identify compensatory movements and how effective they are. Onset of falls is likely. Identify cause of falls and future risk via a structured assessment, taking into consideration the context of the fall as well as frequency. Identify ‘hot spots’ in the home and consider compensatory strategies such as visual cues, carer-prompts. Again, test dual task gait and increase task difficulty. Consider walking aids as mobility impairment increases.
Immobility
The role of the physiotherapist for people with more advanced symptoms is centred on the provision of palliative care and ensuring maximal independence [15]. It is critical to optimise quality of life regardless of life expectancy. Vital to successful physiotherapy at this stage is input from multi-disciplinary team members who are co-ordinating this phase of care. Expertise regards positioning, equipment provision, maintenance of airways, and passive movements/ massage will be required. Focus on functional mobility including bed mobility and transfers/mobility during the night. Maintenance of airways will become a priority. Teach active cycle of breathing techniques,assisted cough, modified postural draining if appropriate, as well as relaxation techniques, with support of a carer. Input is likely to be in a person’s home and centred around the comfort of the individual and needs of them and their families. Liaise with the palliative care team and respiratory home ventilation team as required.
Physiotherapy intervention for MSA-P
As noted above, although signs and symptoms overlap, MSA is characterised by distinct phenotypes. Evidence for MSA-P is inferred from movement science research for PD.
Physiotherapists are encouraged to read the paper by Rochester and colleagues which outlines a strategy for intervention [16], and to familiarise themselves with PD physiotherapy guidelines [17].Use of cognitive strategies is encouraged from early on, and can be a powerful adjunct to intervention [18]. Identify strategies for safe ambulation, such as stepping in confined spaces, turning, and negotiating doorways. Be aware of visuo-spatial deficit which may impact on perception of body in space and relation of body to external objects. People with MSA-P are also likely to experience freezing and festination, for which cues are effective [18] and best taught early on.
Silva-Batista et al (2014) present a case study of the use of resistance training for instability in MSA [28] and Lindop and Skelly (2022) offer a Multidisciplinary Guide to Management of Parkinson’s Disease, including a chapter on Atypical Parkinsonian Syndromes [29].
As noted by Flabeau [9], physiotherapy remains the best therapeutic option for cerebellar ataxia in MSA. There are no guidelines as such for intervention for MSA-C or for degenerative ataxias in general with current evidence reported as ‘Level II’ [19]. A systematic review indicated that effective rehabilitation should be home-based and intensive, and it should start in earlier stages of disease, given the potential for early rather than late gains. Studies concerning degenerative cerebellar ataxias indicate benefits from early, intensive, high-frequency training programme followed by maintenance therapy [20]. Preservation of balance and strength (core strength and large muscle groups) is indicated, especially in the early stages. Ataxia UK have produced guidance for physiotherapists on their website For Physiotherapists – Ataxia, taken from their document ‘Management of the Ataxias towards best clinical practice’ (Ataxia UK, 2016) [26].
Outcome measures
Use of outcome measures may be appropriate but given the deteriorating nature of MSA you may prefer to be guided by the presence and intensity of current symptoms, via self-report. Along with the usual physiotherapy outcomes (timed walk, functional mobility scales, balance and falls self-efficacy etc.), you may wish to use a disease-specific scale such as the Unified MSA Rating Scale (UMSARS) [21]. The UMSARS is a 4-domain scale that includes impairment and functional tasks, motor ability, autonomic function and global disability. Responsiveness has been examined [21,22] and decline in UMSARS scores over 2 years reported. It appears to be more responsive that MSA-specific, health quality of life scales [23].
Key points
- MSA presents as a heterogeneous disorder with often a mix of symptoms.
- Treat early and maintain therapy input over the course of the disease.
- Maintain strength and physiologic fitness within limits of autonomic dysregulation.
- Work with MDT colleagues to provide holistic support to people with MSA.
References
- A Schrag, Y Ben-Shlomo, and NP Quinn Prevalence of progressive supranuclear palsy and multiple system atrophy; a cross sectional study. Lancet. 2006; 354: 1771-1775
- Parkinsons website http://www.parkinsons.org.uk/content/what-parkinsons
- GK Wenning and F Krismer Multiple System Atrophy; Chapter in Oxford Textbook of Movement Disorders 2013 Ed D Burn; Series Ed C Kennard
- PA Hanna, JJ Jankovic, and JB Kirkpatrick Multiple System Atrophy: The putative causative role of environmental toxins. Arch Neuro 199; 56:90 – 4
- L Swan and J Dupont Multiple System Atrophy. Physical Therapy. 1999; 79:488-494.
- S Gilman, PA Low, N Quinn et al. Consensus statement on the diagnosis of multiple system atrophy. J Auton Nerv Syst 1998;74:189–92.
- S Gilman, GK Wenning, et al. Second consensus statement on the diagnosis of multiple system atrophy. Neurology. 2008; 71: 670-676.
- GK Wenning, F Gesser, et al The natural history of Multiple System Atrophy: a prospective European cohort study. Lancet Neurol. 2013; 12: 264-74.
- O Flabeau, G Meissner, and F Tison. Multiple System Atrophy: current and future approaches to management. Therapeutic Advances in Neurological Disorders. 2010; 3(4):249-263.
- AD Holler, JM Tsao, et al Effectiveness of an inpatient movement disorders program for patients with atypical Parkinsonism Parkinsons Dis 2012:871974. doi: 10.1155/2012/871974
- JH Sen, SW Yong, SK Song, et al A Case-Control Study of Multiple System Atrophy in Korean Patients. Movt Disord 2010; 29: 1953-1959.
- J Jain, J Dawson, NP Quinn and ED Playford Occupational therapy in Multiple system Atrophy: A pilot Randomised Controlled Trial Movt Disord 2004; 19 (11): 1360-1364
- E Fonteyn, S Keus, C Verstappen and B van de Warrenburg Physiotherapy in degenerative cerebellar ataxias: Utilisation, patient satisfaction and professional expertise. Cerebellum 2013; 12: 841-7
- FB Horak, DM Wrisley, and J Frank The Balance Evaluation Systems Test (BESTest) to differentiate balance deficits. Phys Ther. 2009; 89: 484-98
- F Lindop, L Brown, M Grazianno, and D Jones. Atypical Parkinsonism: making the case for a neuropalliative rehabilitation approach. International Journal of Therapy & Rehabilitation 2014; 21 (4):176-182
- L Rochester, S Lord and A Nieuwboer (2011) Physiotherapy for Parkinson’s disease: defining evidence within a framework for intervention Neurodegen. Dis. Manage. 1(1), 57–65.
- European Guidelines for physiotherapy in Parkinson’s disease SHJ Keus, M Munneke, M Graziano et al. European Physiotherapy Guideline for Parkinson’s disease. 2014. KNGF/ParkinsonNet (www.ParkinsonNet.info),
- A Nieuwboer et al Cueing training in the home improves gait-related mobility in Parkinson’s disease: the RESCUE trial J Neurol Neurosurg Psychiatry 2007;78:134-140
- B van de Warrenburg et al EFNX/ENS Consensus on the diagnosis and management of chronic ataxias in adulthood E Jnl Neurol 2014
- E Fonteyn, S Keus, C Verstappen, L Schols, I de Groot, and B van de Warrenburg 2013The effectiveness of allied health care in patients with ataxia: A systematic review
- GK Wenning, et al with MSA group Development and validation of the Unified Multiple System Atrophy rating scale Movt Disord 2004; 19 (12):1391-1402
- N Stefanovo, P Bucke, S Duerr, and GK Wenning Multiple System Atrophy: An update Lancet Neurol 2009; 8:1172-8
- WG Meisnner, et al Assessment of quality of life with the Multiple System Atrophy Health-related quality of life scale Mov Disord 2012; 27 (12) 1574-77
- Wenning GK, Stankovic I, Vignatelli L, Fanciulli A, Calandra-Buonaura G, Seppi K, Palma JA, Meissner WG, Krismer F, Berg D, Cortelli P, Freeman R, Halliday G, Höglinger G, Lang A, Ling H, Litvan I, Low P, Miki Y, Panicker J, Pellecchia MT, Quinn N, Sakakibara R, Stamelou M, Tolosa E, Tsuji S, Warner T, Poewe W, Kaufmann H. The Movement Disorder 10.1002/mds.29005. Epub ahead of print. PMID: 35445419
- My Treatment Approach to Multiple System Atrophy (mayoclinicproceedings.org) accessed October 2022. Full reference My Treatment Approach to Multiple System Atrophy – Mayo Clinic Proceedings
- Ataxia UK 2016 ‘Management of the ataxias towards best clinical practice’ Ataxi UK Medical Guidelines. Third Edition. v3m Dec 2016 – updated Sep 2019.pdf
- Ataxia UK website accessed Oct 2022 For Physiotherapists – Ataxia
- Silva-Batista C, Kanegusuku H, Roschel H, Souza EO, Cunha TF, Laurentino GC, Manoel N Jr, De Mello MT, Piemonte ME, Brum PC, Forjaz CL, Tricoli V, Ugrinowitsch C. Resistance training with instability in multiple system atrophy: a case report. J Sports Sci Med. 2014 Sep 1;13(3):597-603. PMID: 25177187; PMCID: PMC4126297.
- Lindop, F and Skelly, R (eds) 2022. Parkinson’s Disease: A multidisciplinary guide to management. UK:Elsevier
Further reading
- The Multiple System Atrophy Trust website http://www.msatrust.org.uk/
- Parkinson’s UK www.parkinsons.org.uk/
- Association of Physiotherapists in Parkinson’s disease Europe
- BCP Ghosh Multiple System Atrophy British Journal of Neuroscience Nursing 2014; 10, (3); 112 -115
- MSA research networks in Europe , the USA and Japan
(All websites accessed 18/09/2014)
- Raccagni et al, Physiotherapy improves motor function in patients with the Parkinson variant of multiple system atrophy: A prospective trial.
https://www.prd-journal.com/article/S1353-8020(19)30417-1/fulltext
This Guide was compiled by Dr Sue Lord and Mrs Dadirayi Mhiripiri, Institute for Neuroscience, Newcastle University, UK. Many thanks to Vicky Carr, Fiona Lindop, Sarah Hornsby, Samantha Pavey, Jill Lyons and Katie Rigg for expert advice and assistance with text. Updated in February 2023, with thanks to Jade Donnelly, Physiotherapist.